- Home
- About
- Contact
- Upgrade to microsoft office 2016
- J unertl scope for sale
- Abbyy lingvo x3
- Surface trimble business center
- Commandos 2 men of courage keeps freezing and black screen
- Tune the guitar
- Naruto shippuden ultimate ninja 5 cheats pcsx2
- Tiger woods pga tour 2003 rom
- My kindle books
- Coh opposing fronts steam cd key
- Factoring trinomials worksheet a-1
- Home depot planogram portal
- Report efilm lite
- Download clamxav for mac os x
- Tokyo species 2012 download
- Rumpus meaning
- Xps peak data
- Portable electronic workbench
- Artificial academy 2 windows 10 download
- Red dead redemption pc g2a
- Endwell auto lab
- Nocash gba emulator crashing
- How to find p value for hypothesis test calculator
- Digimoto 5
- Typing master pro serials latest 2018
- G930w8 unlock modem file
- 3d movies for vr goggles

#XPS PEAK DATA HOW TO#
Differentiated carbon Auger and D-Value plot for various carbons References X-ray Photoelectron Spectroscopy (XPS) Sources of Information Principles of XPS and Auger How to prepare samples for XPS Instrumentation, X rays, Photoelectron detection Data acquisition Quantitative and Qualitative analyses Spin-orbit splitting, Plasmons, Shake-up, etc. By recording the carbon x-ray induced Auger peak and taking the maxima and minima from its differential form (figure 1), then the sp2% may be found. SP2 carbon ~ 284.0 – 284.5 eV (HOPG typically seen at 284.5 eV )īy far the best way of determining sp2/sp3 ratio is by means of the D-value.Organic/Polymeric materials (these are average values – See reference ): Typically there will be some C-O and C=O structure associated (286 – 288 eV), however depending no the nature of the material, this is not always a good reference despite its wide use – for example carbide materials or for aluminium, the carbon is typically around 286 eV.In fact the majority of samples exposed to the atmosphere will have a considerable concentration of carbon contamination ~ 2 nm thick.Īdventitious Carbon: 284.8 eV (C-C and C-H bonds). Equally it may be bound to O, S, N, metals in different ratios or be present as a carbide phase. An XPS spectra is created by plotting the number of electrons verses their binding energy.
#XPS PEAK DATA FULL#
Oxygen is a highly reactive element that promotes rapid combustion and is often used in industrial application.Carbon, in its many forms may exhibit full sp2 or sp3 hybridisation, or a combination. Oxygen is colorless, odorless and tasteless in its gaseous form, and condenses to pale blue liquid and solid forms. Accounting for one fifth of the earth’s atmosphere, oxygen combines with most elements and is a component of thousands of organic compounds. Oxygen is critical for life on Earth, produced by plants during photosynthesis and necessary for aerobic respiration in animals. This video shows how to analyze XPS data, peak position & survey scan easily. Oxygen, the “elixir of life,” was discovered by Joseph Priestly and Carl Wilhem Scheele independently of each other in the 1770’s. This tutorial is for all who have performed XPS and analysing data.
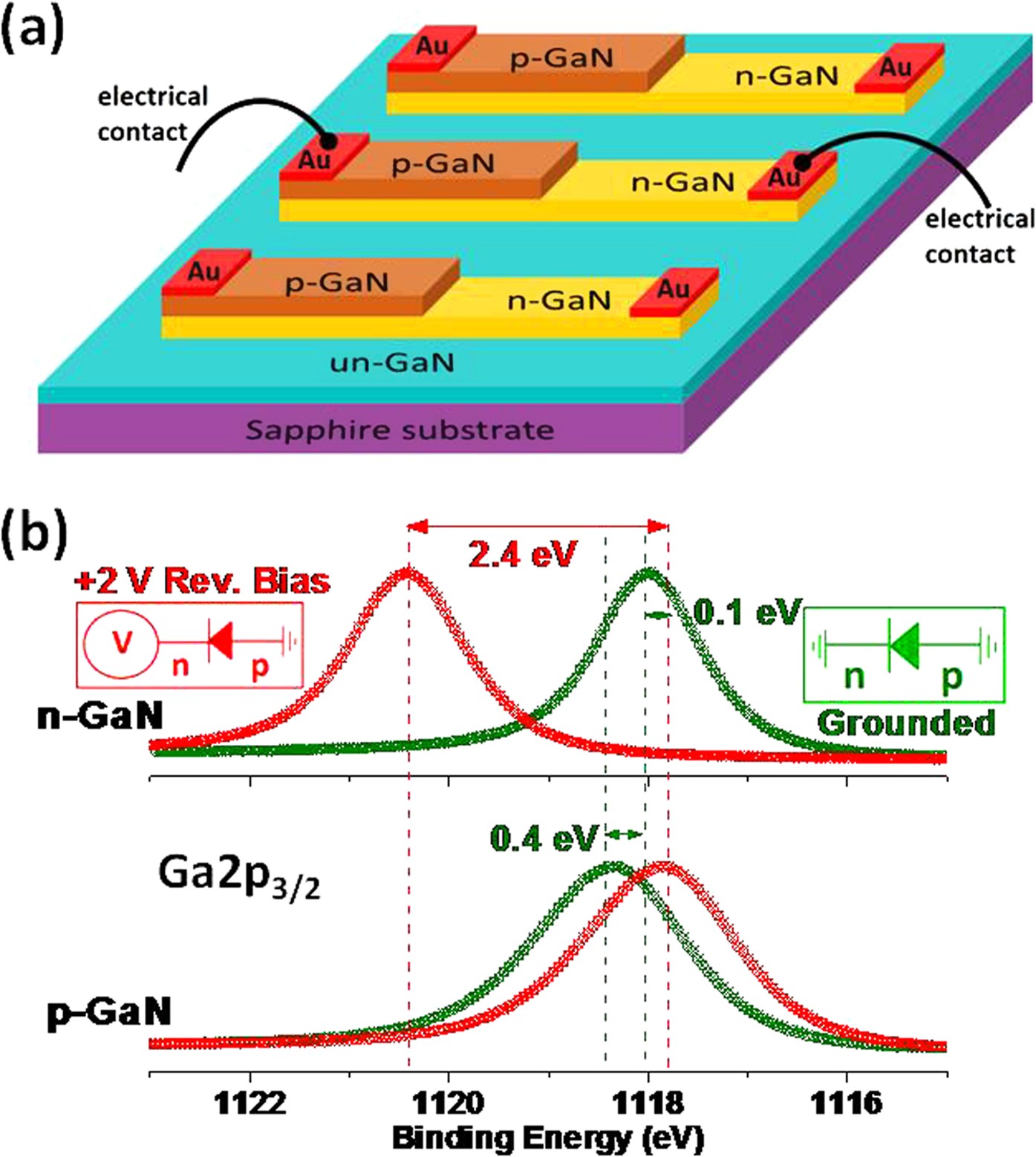
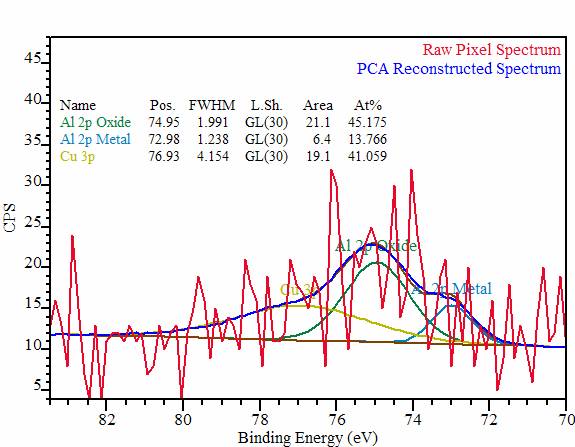
Nohira et al., Journal of Non-Crystalline Solids 303 (2002) 83–87 Data are typically collected in a) Survey mode to obtain the complete inventory of elements on a material surface, and b) high resolution scans of peaks of interest to reveal the bound state (chemical bonds) involving. measured photoelectron counts on the Y-axis.

